At Sun Nuclear we are committed to agility and dedicated to innovation - it's what drives our fast-paced, customer-centric culture, and our focus on continuous improvement in pursuit of Patient Safety. See current openings.

Patient Safety Starts Here
The pursuit of Patient Safety is at the core of what we do.
We develop and deliver Independent, Integrated Quality Management solutions for Radiation Therapy, Diagnostic Imaging and Patient Alignment.
We believe Independence and Integration are equally critical to achieving meaningful Quality Management for today's hospitals, cancer centers and healthcare networks.
If Patient Safety matters to you, Sun Nuclear may be the right environment for you.
Our Values
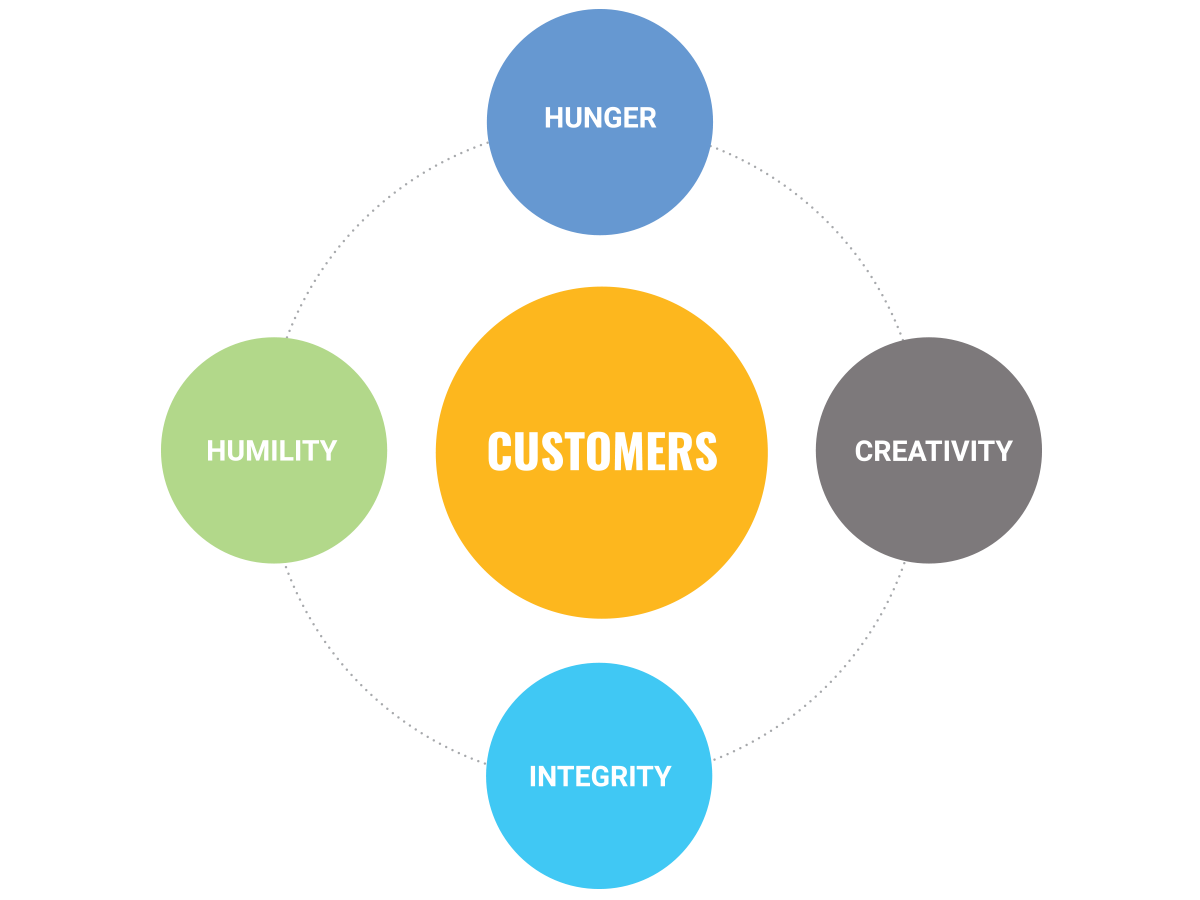
Customers
We solve problems for our customers in ways that are smart and innovative.
Humility
We are continuously learning and stay open to new ideas.
Hunger
Our drive to offer highly valuable solutions pushes us to challenge ourselves.
Creativity
We question assumptions and take risks in search of better, more efficient ways of doing things.
Integrity
We hold ourselves accountable to the highest level, and take pride in an unwavering commitment to exceptional service.
Join the leader in Quality Management solutions.
Benefits

Health & Welfare
- Medical / Prescription Coverage /Telemedicine
- Dental and Vision Coverage
- Health Savings Account with Employer Contribution
- Dependent Care Spending Account
- Basic Life Insurance and Accidental Life & Dismemberment Insurance
- Additional Life and Dependent Life Insurance available
- Paid Short Term and Long-term Disability
- Voluntary Benefits - Cancer, Critical Illness, Accident and Hospital Plans
- Employee Assistance Program (EAP)
- Travel Assistance Program

Retirement Planning
- 401(K) Plan to prepare for retirement
- Employer match, flexible based on employee denominations
- Access to retirement professionals for financial education and consultation
- 24/7 online access to account information

Life & Work Balance
- Flexible schedules - including early or late start, and/or regular time off
- Remote work - work from home some or all of the time
- Paid Time Off (PTO) - start with a PTO allotment that accrues over time
- Paid Holidays - enjoy eight paid holidays per year
- Maternity, Paternity & Adoption Leave - take time to care for a new addition to your family
- Family Care Leave & Qualifying Need Leave - leave for when you need it
Benefits dependent on hiring agreement and management approval.
"It really is a family culture, a tight knit group that works closely together, has a lot of fun while we do it. Also, we truly are a team that takes a lot of pride in what we do and who we are doing it for."
Melbourne, FL USA

Apply Now
Inspired to join an industry leader making a genuine difference? Sun Nuclear is the place for you.
Send Us a Resumé
We are always looking for talented applicants. If none of our current openings are a good fit, email us your resumé to be considered for future opportunities.
Sun Nuclear is an equal opportunity employer and does not discriminate on the basis of race, color, religion, sex, sexual orientation, gender identity, national origin, pregnancy, age, disability, veteran status, genetic information or other protected status.
Applicants with disabilities may be entitled to reasonable accommodation under the terms of the Americans with Disabilities Act and certain state or local laws. A reasonable accommodation is a change in the way things are normally done which will ensure an equal employment opportunity without imposing undue hardship on the company.
If you require assistance completing this application due to a disability, please contact [email protected] or call 321-259-6862 to ask for an accommodation or an alternative application process.